 :max_bytes(150000):strip_icc()/GettyImages-646181145-57bda5b13df78c8763e7e99d.jpg)
The region from 8 to 15 microns (µm) is referred to by Earth scientists as thermal infrared since these wavelengths are best for studying. The electromagnetic spectrum covers electromagnetic waves with frequencies ranging from below one hertz to above 10 25 hertz, corresponding to wavelengths from thousands of kilometers down to a fraction of the size of an atomic nucleus. This region of the spectrum is divided into near-, mid-, and far-infrared. Each of these energy packets is known as a quantum (plural: quanta). A remote control uses light waves just beyond the visible spectrum of lightinfrared light wavesto change channels on your TV. Visible light range comes under the range of EM spectrum between infrared and ultraviolet, having a frequency of about 4 x 10 14 to 8 x 10 14 cycles per second. The reality, which Planck discovered, is that energy is not continuous but quantized-meaning that it can only be transferred in individual “packets” (or particles) of the size h ν h\nu h ν h, \nu. This was a shocking discovery, because it challenged the idea that energy was continuous, and could be transferred in any amount. Note that the visible part of the spectrum, the only type of electromagnetic radiation. Human eyes can detect and respond to only a tiny part of the electromagnetic spectrum. Read on to learn more about the range, frequency, and properties. 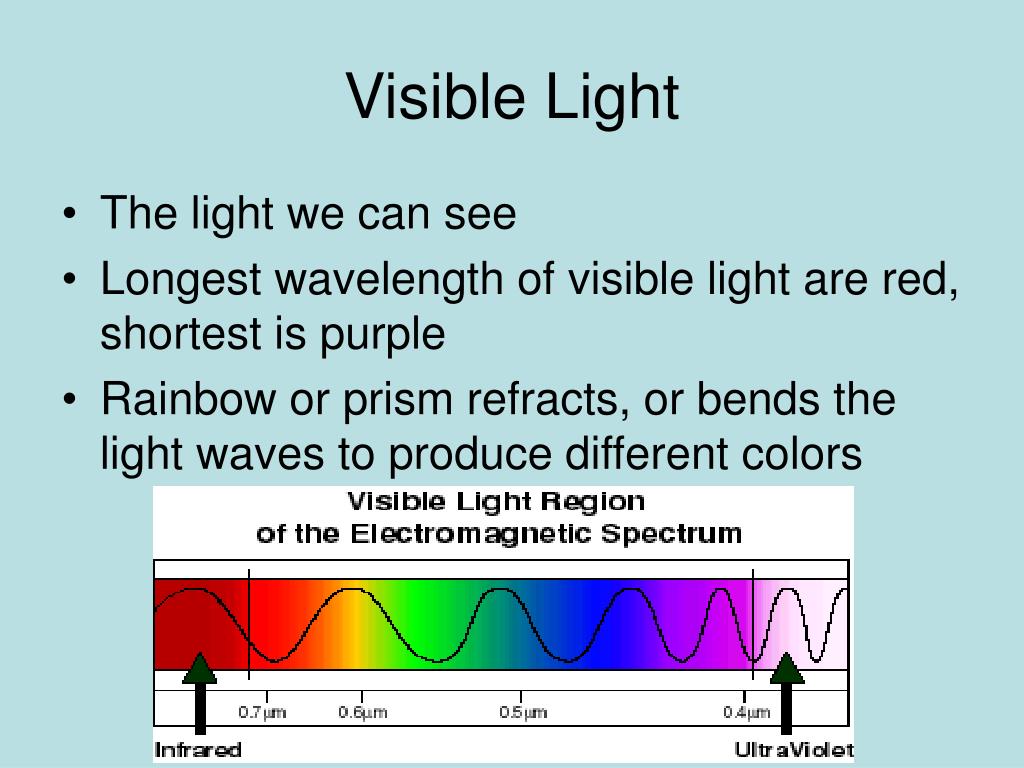
6 2 6 × 1 0 − 3 4 J ⋅ s 6, point, 626, times, 10, start superscript, minus, 34, end superscript, start text, space, J, end text, dot, start text, s, end text, and ν \nu ν \nu is the frequency of the light absorbed or emitted. The image below shows the names given to different regions of the EMS. The visible light spectrum is a segment of the electromagnetic spectrum, which makes bodies and objects visible to our eyes. Planck observed that matter actually absorbed or emitted energy only in whole-number multiples of the value h ν h\nu h ν h, \nu, where h h h h is Planck’s constant, 6.626 × 1 0 − 34 J ⋅ s 6.626\times10^ 6. A spectrum (plural spectra or spectrums) is a condition that is not limited to a specific set of values but can vary, without gaps, across a continuum.The word was first used scientifically in optics to describe the rainbow of colors in visible light after passing through a prism. 
Planck found that the electromagnetic radiation emitted by blackbodies could not be explained by classical physics, which postulated that matter could absorb or emit any quantity of electromagnetic radiation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
